In cost-effectiveness analysis, we often rely on the Incremental Net Benefit (INB) to determine whether one treatment is more cost-effective than another. As discussed earlier, a positive value of ![]() suggests that treatment 1 is preferred over treatment 2 at a specific willingness-to-pay (WTP) threshold
suggests that treatment 1 is preferred over treatment 2 at a specific willingness-to-pay (WTP) threshold ![]() . However,
. However, ![]() is not directly known. It depends both on the population parameters, such as the mean costs and effects, and the chosen value of
is not directly known. It depends both on the population parameters, such as the mean costs and effects, and the chosen value of ![]() . To address this uncertainty, we estimate
. To address this uncertainty, we estimate ![]() from sample data and then calculate the probability that this estimate is greater than zero. This probability, when plotted across different values of
from sample data and then calculate the probability that this estimate is greater than zero. This probability, when plotted across different values of ![]() , forms the Cost-Effectiveness Acceptability Curve (CEAC).
, forms the Cost-Effectiveness Acceptability Curve (CEAC).
Defining the CEAC
The CEAC essentially shows the likelihood that a treatment is cost-effective for a range of WTP values. Let us assume we are comparing two treatments in a randomized controlled trial (RCT). For each treatment, we have cost and effectiveness data:
 and
and  represent the costs of treatment 1 and 2,
represent the costs of treatment 1 and 2,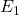 and
and  represent their effectiveness.
represent their effectiveness.
The INB is given by:
![]()
To construct a CEAC, we replace the population means ![]() with their corresponding sample means. The resulting estimator,
with their corresponding sample means. The resulting estimator, ![]() , is approximately normally distributed if certain assumptions are met. The CEAC is then defined as:
, is approximately normally distributed if certain assumptions are met. The CEAC is then defined as:
![]()
plotted against various values of ![]() .
.
When Normality Holds
If cost and effectiveness data follow a bivariate normal distribution, the calculation of CEAC is straightforward. The distribution of ![]() can be derived, and we can compute the probability
can be derived, and we can compute the probability ![]() using the normal distribution. This makes it easy to visualize how the probability of cost-effectiveness changes as
using the normal distribution. This makes it easy to visualize how the probability of cost-effectiveness changes as ![]() increases.
increases.
For example, suppose:
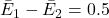 QALYs,
QALYs,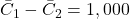 USD.
USD.
If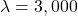 USD per QALY, the INB estimate is:
USD per QALY, the INB estimate is:
![]()
Since this value is positive, the probability that treatment 1 is cost-effective would be high. By calculating this probability for a range of ![]() values, we create the CEAC curve.
values, we create the CEAC curve.
When Normality Does Not Hold
Real-world data often deviates from normality due to skewed costs or irregular distributions of effectiveness. In such cases, calculating the CEAC directly becomes challenging. Instead, we use asymptotic approximations or bootstrap methods. Bootstrapping involves repeatedly resampling the data to generate a distribution of ![]() and then estimating the proportion of times it is positive. This approach does not rely on strict distributional assumptions and is widely used in health economics.
and then estimating the proportion of times it is positive. This approach does not rely on strict distributional assumptions and is widely used in health economics.
For example, imagine a dataset of 1,000 patients split into two treatment groups. By resampling with replacement and calculating ![]() for each resample, we can build a probability curve that reflects the variability in our data.
for each resample, we can build a probability curve that reflects the variability in our data.
What is CEAF?
A related concept to CEAC is the Cost-Effectiveness Acceptability Frontier (CEAF). While CEAC shows the probability that a single treatment is cost-effective, CEAF identifies which treatment, among all options, has the highest net benefit for a given value of ![]() . In simpler terms, CEAF provides a summary curve that tells decision-makers which treatment is most likely optimal as willingness-to-pay changes.
. In simpler terms, CEAF provides a summary curve that tells decision-makers which treatment is most likely optimal as willingness-to-pay changes.
Mathematically, CEAF is computed as the probability that the treatment with the maximum ![]() is cost-effective:
is cost-effective:
![]()
where ![]() represents the treatments being compared.
represents the treatments being compared.
Interpreting CEAC and CEAF in Decision-Making
Both CEAC and CEAF are decision-support tools. For example, a CEAC might show that treatment 1 has a 70% probability of being cost-effective when ![]() USD per QALY. If this probability is higher than other treatments, it strengthens the case for choosing treatment 1. CEAF, on the other hand, shows the treatment with the highest probability of being cost-effective at each threshold. For pharma executives, these curves help determine price positioning and payer negotiation strategies.
USD per QALY. If this probability is higher than other treatments, it strengthens the case for choosing treatment 1. CEAF, on the other hand, shows the treatment with the highest probability of being cost-effective at each threshold. For pharma executives, these curves help determine price positioning and payer negotiation strategies.
Lets look at an Example
Let us consider two cancer drugs being compared in a clinical trial:
- Drug A: Cost = $20,000, Effectiveness = 3.5 QALYs.
- Drug B: Cost = $15,000, Effectiveness = 3 QALYs.
The INB, for ![]() , is:
, is:![]()
A positive INB suggests Drug A is cost-effective. By varying ![]() and repeating this calculation, we can plot a CEAC showing how confidence in Drug A’s cost-effectiveness changes with different WTP thresholds.
and repeating this calculation, we can plot a CEAC showing how confidence in Drug A’s cost-effectiveness changes with different WTP thresholds.