In many cancers today, some patients never experience the event you are modeling. They don’t relapse. They don’t progress. They don’t die from the disease during long follow-up. Clinically, we often say they are functionally cured.
Traditional survival models are not built for this situation. They quietly assume that if you wait long enough, everyone will eventually fail. That assumption used to be reasonable. In modern oncology, it often isn’t.
Cure models exist because that assumption breaks reality.
The core idea of cure models
A cure model does one simple but powerful thing: it separates the population into two latent groups.
One group is cured. These patients will never experience the event of interest. The other group is uncured. These patients remain at risk over time. You don’t directly observe which group a patient belongs to. The model estimates this probabilistically.
This distinction matters enormously for HEOR, because long-term survival shapes life-years, QALYs, costs, and ICERs.
Why standard survival models fail here
Suppose you have 10 years of follow-up data in oncology. After year 6, the survival curve flattens. Almost no additional events occur. A Weibull or exponential model will still push the curve downward eventually. It has no way to say, “Some people are done with risk.”
This leads to:
- Underestimated long-term survival
- Underestimated QALYs
- Overestimated ICERs
Cure models fix this by explicitly modeling a plateau.
Mixture cure models (the intuitive version)
The mixture cure model assumes that your population is a mixture of cured and uncured patients.
The survival function is written as:
![]()
Here:
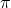 is the cure fraction.
is the cure fraction.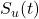 is the survival function of the uncured group.
is the survival function of the uncured group.
Interpret this carefully.
A proportion ![]() of patients survive forever with respect to the event. The remaining proportion
of patients survive forever with respect to the event. The remaining proportion ![]() follow a standard survival distribution.
follow a standard survival distribution.
This is a very natural way to think about cancer outcomes.
What the curve looks like
Early in time, survival drops as uncured patients experience events. Later, the curve flattens and approaches ![]() . That flat tail is not a modeling artifact. It is the estimated cured proportion.
. That flat tail is not a modeling artifact. It is the estimated cured proportion.
HTA reviewers often find this interpretation intuitive because it aligns with clinical thinking.
Adding covariates to the cure fraction
The cure fraction does not have to be the same for everyone.
You can model it using logistic regression:
![]()
This allows treatment, biomarkers, or patient characteristics to affect the probability of being cured, not just time to event.
This distinction is extremely important. A treatment might not slow progression much in uncured patients, but dramatically increase the chance of cure. Traditional survival models cannot express this difference. Cure models can.
The uncured survival component
For uncured patients, you still need a survival model. This can be Weibull, log-normal, or even flexible parametric.
For example:
![]()
The full model then combines a binary latent process (cured vs uncured) and a continuous time-to-event process for uncured patients. That’s why cure models are sometimes described as two-part survival models.
Non-mixture cure models (more subtle, same goal)
Non-mixture cure models approach the same problem differently. Instead of mixing two survival curves, they model the cumulative hazard so that it levels off over time.
A common form is:
![]()
with
![]()
If the cumulative hazard reaches a finite limit, survival never goes to zero. The plateau represents cure. There is no explicit cured subgroup. Instead, cure emerges mathematically from the hazard structure.
How mixture vs non-mixture differ conceptually
Mixture models say: some patients are cured. Others are not. Non-mixture models say: risk fades away completely for some patients over time. Both produce survival plateaus. The choice is often pragmatic. Mixture models are easier to explain, while non-mixture models can be more stable numerically. In HEOR, interpretability often matters more than elegance.
Let’s look at an oncology example
Imagine 800 patients with metastatic cancer. Half receive standard care. Half receive a new immunotherapy.
After 5 years, standard care survival stabilizes at 10%, while immunotherapy survival stabilizes at 30%.
A cure model estimates:
![]()
![]()
Among uncured patients, time to progression is similar across arms. This tells a very specific story: the new therapy does not merely delay events. It creates long-term survivors. This distinction is critical for value assessment.
Why cure models are powerful
From a health economics perspective, cure models do three things extremely well. First, they produce realistic long-term survival when plateaus exist. Second, they separate early hazard reduction from long-term benefit, which aligns with modern oncology mechanisms. Third, they prevent artificial downward drift in survival that inflates ICERs unfairly.
Cure models and cost-effectiveness
Survival directly affects expected effectiveness:
![]()
Those life-years flow into QALYs and ICERs:
![]()
If you ignore cure, you undercount ![]() . If you overestimate cure, you overstate value.
. If you overestimate cure, you overstate value.
Cure models don’t remove uncertainty — they make it explicit.
When cure models are appropriate
They are not always justified. Cure models are most appropriate when Kaplan–Meier curves show a clear plateau, follow-up is long enough to observe stabilization, and clinical experts agree cure is plausible. Using cure models without evidence is risky. But avoiding them when evidence exists is equally problematic.
Final to remember
A cure model asks a different question than most survival models. Not just: “How long until the event?” But: “Who is ever going to experience the event at all?” In modern oncology HEOR, that question often matters more.